The global tenodesis screw market is experiencing a transformative era, driven by the increasing incidence of sports-related injuries and a rising aging population seeking active lifestyles. As a cornerstone of sports medicine, tenodesis screws—specifically interference screws—play a critical role in ACL reconstruction, biceps tenodesis, and ligament repairs. The shift from traditional titanium to bio-absorbable materials and PEEK (Polyetheretherketone) has redefined surgical outcomes, offering better post-operative imaging and reduced long-term complications.

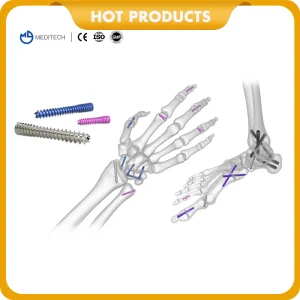
Our flagship tenodesis solutions include the Interference Screws for ACL Reconstruction. Engineered for maximum pull-out strength and biological compatibility, these screws are available in both PEEK and medical-grade titanium. They provide stable fixation for both soft tissue and bone-tendon-bone (BTB) grafts.
The cannulated design allows for precise placement over a guide wire, minimizing surgical error and reducing time in the operating room. Whether for professional athletes or recreational enthusiasts, our screws ensure a return to peak performance.
Explore Technical Specifications →
The move toward Bio-composite materials like HA (Hydroxyapatite) infused PEEK is enhancing bone integration, making the implant a dynamic part of the healing process rather than just a mechanical fix.
Modern tenodesis procedures prioritize smaller incisions. Our screws are designed for arthroscopic delivery, reducing scarring and recovery time for patients globally.
Integration with robotic-assisted surgery and 3D preoperative planning is demanding tighter tolerances in screw manufacturing, a challenge our factory meets with CNC precision.
From private clinics in Brazil to major hospital networks in Germany, the demand for high-quality, cost-effective tenodesis screws is universal. Procurement managers today look for more than just a product; they seek a supply chain partner capable of providing:
Local Application Scenario: In high-altitude regions or sports-intensive locales like the Alps or North American ski resorts, orthopedic centers require robust stock of interference screws to handle the seasonal surge in ligament injuries.
China has evolved from a low-cost manufacturing hub into a leader in precision medical engineering. Our facility combines cost-efficiency with cutting-edge R&D, offering significant advantages:
Featured: Titanium Headless Compression Screws
The company is committed to producing and selling advanced medical devices, equipment, and disposable medical supplies. Our product range primarily includes the production and sale of instruments for otolaryngology, orthopedics, urology, abdominal surgery, gynecology, and related disposable equipment, as well as sports and fitness equipment. The main focus is on orthopedic devices: implants, mechanical equipment, etc.
Why choose Pure? Our company specializes in the production and sale of medical devices, and we pride ourselves on our commitment to excellence in every aspect of our business. We operate from a 3000+ Square Meters built facility with multiple production lines and an extensive export network spanning 20+ countries.
Tenodesis screws are engineered devices used primarily in the reattachment of tendons to bone. This procedure is common in the shoulder (biceps tenodesis) and the knee (ACL/PCL reconstruction). The success of these surgeries depends heavily on the "initial fixation strength"—the ability of the screw to hold the tendon in place against physiological loads until biological healing occurs.
PEEK (Polyetheretherketone) has become the material of choice for many surgeons due to its radiolucency. Unlike titanium, PEEK does not create "artifacts" on MRI or CT scans, allowing doctors to clearly monitor the healing process. Furthermore, PEEK has a modulus of elasticity similar to human bone, which can reduce stress shielding.
Titanium Alloys (Ti6Al4V) remain the gold standard for high-torque applications. Their superior biocompatibility and the ability to undergo osseointegration make them ideal for complex reconstructions where mechanical failure is a high risk.
Our production process involves multiple stages of quality control. It begins with the selection of raw materials, followed by precision CNC machining. Each tenodesis screw undergoes a rigorous cleaning process, often involving ultrasonic baths, to ensure it is free of any manufacturing residues. For sterile-packed products, we utilize Gamma irradiation or EO sterilization, meeting international medical standards.
As a leading exporter, we understand the nuances of international trade in medical devices. We provide comprehensive documentation, including Certificates of Analysis (CoA) and compliance records, ensuring smooth customs clearance and hospital acceptance across Europe, the Americas, and Southeast Asia. Our technical team is always available to discuss custom thread designs or specific material requirements for your local market.