
Korea Standard Titanium Orthopedic Implants Fixation Surgical Cannulated Screw
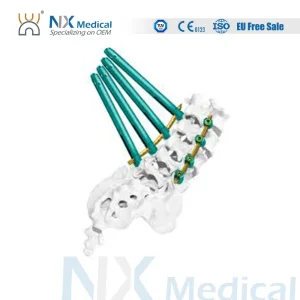
Seoul Series Orthopedic Implant Cannulated Bone Cement Spinal Pedicle Screw 5.5mm System

Korea Precision Titanium Scew, Headless Screw, Compression Screws, Cannulated Screw

High-Performance Hb7.3 Titanium or Ss Cannulated Compression Screws
The Cannulated Screws Factories in Korea represent the pinnacle of medical device engineering in the Asia-Pacific region. As a global hub for "K-Medical" innovation, South Korea has transformed its orthopedic manufacturing sector into a high-tech powerhouse. The industry is not merely about production; it is about integrating AI-driven design, robotic manufacturing, and advanced metallurgy to create implants that redefine patient outcomes.
The demand for cannulated screws in South Korea is driven by two primary factors: a rapidly aging population requiring spinal and joint interventions, and a booming sports medicine sector. As the "Golf Capital" of Asia, Korea sees a high frequency of sports-related injuries, leading to a sophisticated market for interference screws for ACL reconstruction and headless compression screws for small bone fractures.
Korean factories utilize digital twin technology to simulate the stress load on cannulated screws before they leave the production line, ensuring zero-fail performance in spinal surgeries.
Beyond Grade 5 Titanium, Korean innovators are exploring PEEK and bio-absorbable magnesium alloys to reduce the need for secondary hardware removal surgeries.
Strategically located near Incheon, our manufacturing facilities ensure that surgical implants can be delivered to hospitals in Southeast Asia, Europe, and North America within record timeframes.
When searching for a factory in Korea, global buyers look for the "K-Quality" mark. This involves not only the screw itself but the entire delivery system, including bone-cement injectable technology for osteoporotic patients—a specialty where Korean surgeons and manufacturers have collaborated to lead the world.
The precision of a Cannulated Bone Cement Pedicle Screw manufactured in Korea is unmatched due to the specialized CNC machining processes used in Seoul's tech districts. These screws allow for the guided placement of K-wires, ensuring that surgeons can perform minimally invasive surgeries (MIS) with extreme accuracy. In the context of the 5.5mm system, Korean designs offer superior thread-purchase and pull-out strength, which is critical for the dense demographic of elderly patients with lower bone mineral density.
Factory Insight: Our company is committed to producing and selling advanced medical devices, equipment, and disposable medical supplies. Our product range primarily includes the production and sale of instruments for otolaryngology, orthopedics, urology, abdominal surgery, gynecology, and related disposable equipment, as well as sports and fitness equipment. The main focus is on orthopedic devices: implants, mechanical equipment, etc.
Our team of experts is dedicated to ensuring that our products meet the highest standards of quality and safety. We pride ourselves on our commitment to excellence in every aspect of our business, from the sourcing of raw medical-grade titanium to the final sterilization packaging.

The global orthopedic implant market is shifting toward personalization. In Korea, this trend is manifesting as 3D-printed cannulated screws tailored to the specific anatomical dimensions of a patient's vertebra or femur. By utilizing CT-scan data, factories can now adjust the pitch and length of the screw threads to optimize fixation in compromised bone structures.
Another major trend is the integration of Smart Coatings. Research and development teams in Busan are currently testing hydroxyapatite-coated screws that promote faster osseointegration, effectively turning the metal implant into a "living" part of the patient's skeletal system. This is particularly relevant for the "Active Senior" population in Korea, who refuse to let age slow down their lifestyle.
Furthermore, the MIS (Minimally Invasive Surgery) trend is dictating factory output. Smaller, more robust cannulated headless compression screws are being produced to support foot and ankle surgeries, which are seeing a double-digit growth rate in Korean orthopedic clinics. The ability of Korean factories to pivot quickly to these market demands is what sets them apart from traditional manufacturing giants.
Environmental sustainability is also reaching the factory floor. Leading Korean manufacturers are implementing "Green Factory" initiatives, reducing carbon footprints during the titanium smelting and milling processes. Choosing a Korean partner means supporting a future-ready, ethical supply chain that values both patient health and global environmental standards.
Explore our full range of Korean-engineered orthopedic solutions

Interference Screws for Acl Reconstruction Peek and Cannulated Screws Sports Medicine

Cannulated Headless Compression Screw for Orthopedic Implant

Nx Medical Orthopedic Implant Cannulated Conical Headless Compression Screw - II Metallic

Orthopedic Implant Cannulated Headless Compression Screw - II Metallic Bone Screws
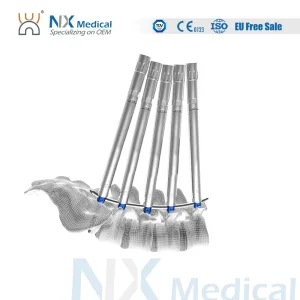
Nx Medical Spine Implant Cannulated Bone Cement Pedicle Screw for Spine Deformity

Orthopedic Cannulated Screw for Bone Fixation Trauma Implant
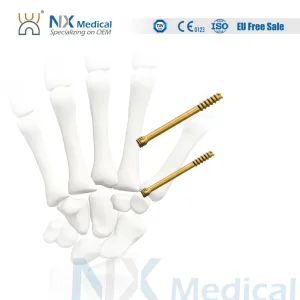
Orthopedic Implant Cannulated Headless Compression Screw Bone Screw Fixation

Factory Direct Supply Orthopedic Spine Implant Bone Cement-Injectable Cannulated Pedicle Screws
Navigating the global medical supply chain requires a partner who understands the nuances of orthopedic surgery and the engineering requirements of different bone types. At Pure, we specialize in bridging the gap between high-level Korean manufacturing and the needs of hospitals worldwide. Whether it's our Urology Instruments, Laparoscopic sets, or our flagship Cannulated Screw systems, our commitment to excellence remains unwavering.
Our "3 Production Lines" are optimized for high-volume output without sacrificing the micron-level precision required for spinal implants. By serving over 20+ exporting countries, we have developed a deep understanding of varied regulatory landscapes, ensuring that our products arrive ready for immediate clinical use.
In conclusion, the Cannulated Screws market in Korea is more than just a manufacturing sector; it is a vital part of the global healthcare ecosystem. By choosing our factory-direct supplies, you are investing in safety, innovation, and the future of orthopedic recovery.
Ready to upgrade your surgical inventory with Korea's finest orthopedic implants?
Send Inquiry Now