 Xc Medico Orthopedic 6.0mm System Spinal Pedicle Screw System Surgical Instrument Set
Xc Medico Orthopedic 6.0mm System Spinal Pedicle Screw System Surgical Instrument Set
 Orthopedic Surgical Instrument Spinal Fixation System 6.0 mm Pedicle Screw Instruments Set
Orthopedic Surgical Instrument Spinal Fixation System 6.0 mm Pedicle Screw Instruments Set
 High-Strength Cocrmo Thoracolumbar Pedicle Screw and Rod System
High-Strength Cocrmo Thoracolumbar Pedicle Screw and Rod System
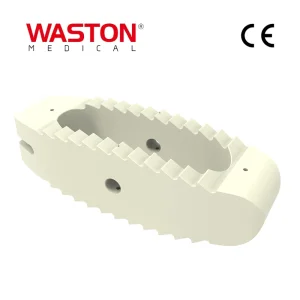 Disposable Surgical Neulen Laminoplasty Thread Inter Reduction Pedicle Screw Orthopaedic Instrument Implant O-Max Cage System
Disposable Surgical Neulen Laminoplasty Thread Inter Reduction Pedicle Screw Orthopaedic Instrument Implant O-Max Cage System
 Spine Surgical Pedicle Screw Titanium Surgery Orthopedic Implants Posterior Thoracolumbar Spinal Fixation System
Spine Surgical Pedicle Screw Titanium Surgery Orthopedic Implants Posterior Thoracolumbar Spinal Fixation System
 High Quality Cannulated Axial-Rotation Pedicle Screw Human Surgical Instrument Orthopaedic Implant Posterior Spinal Internal Fixation Minimally Invasive System
High Quality Cannulated Axial-Rotation Pedicle Screw Human Surgical Instrument Orthopaedic Implant Posterior Spinal Internal Fixation Minimally Invasive System
 Disposable Surgical Neulen Laminoplasty Inter Pedicle Screw Orthopaedic Instrument Bone Graft Implant O-Max Cage System
Disposable Surgical Neulen Laminoplasty Inter Pedicle Screw Orthopaedic Instrument Bone Graft Implant O-Max Cage System
 Sacrual Self-Breaking Plug Titanium Pedicle Screw Surgical Instrument Orthopaedic Implant Posterior Spinal Internal Fixation 5.5 System
Sacrual Self-Breaking Plug Titanium Pedicle Screw Surgical Instrument Orthopaedic Implant Posterior Spinal Internal Fixation 5.5 System
The global spinal surgery market is undergoing a significant transformation, with Pedicle Screw Systems remaining the cornerstone of posterior spinal fixation. As a leading Pedicle Screw Systems supplier and exporter, we have observed a consistent rise in demand driven by an aging global population, the increasing prevalence of degenerative disc diseases, and a surge in spinal trauma cases from sports and industrial accidents.
Currently, the market is characterized by a shift toward value-based healthcare. Hospitals in North America and Europe are prioritizing implants that offer long-term stability and reduced revision rates, while emerging markets in Asia-Pacific and Latin America are seeking cost-effective yet high-quality surgical solutions. This dual demand has positioned Chinese manufacturers as pivotal players in the global supply chain, offering high-precision titanium implants that meet stringent international standards like CE and ISO 13485.
Annual Market CAGR
Global Hospital Partnerships
Medical Grade Materials
Export Destinations
From a commercial perspective, the integration of 3D printing technology and AI-assisted surgical planning is redefining product portfolios. Suppliers are no longer just selling screws; they are providing comprehensive "Spinal Fixation Ecosystems" that include rods, connectors, cross-links, and specialized instrumentation sets. Our role as a factory involves bridging the gap between high-end engineering and clinical accessibility.
The most prominent trend is the move toward minimally invasive techniques. Traditional open surgeries are being replaced by procedures that require smaller incisions, leading to faster patient recovery and less blood loss. This has led to the development of Cannulated Pedicle Screw Systems, which allow for percutaneous placement over a guide wire.
While Titanium alloys (Ti6Al4V) remain the gold standard due to their biocompatibility and MRI compatibility, there is a growing interest in Cobalt-Chrome (CoCrMo) rods for complex deformity corrections where high stiffness is required. Our product line includes both materials to cater to varied clinical indications.
Furthermore, Surface Modification Technology is becoming a competitive edge. Bioactive coatings such as Hydroxyapatite (HA) are being applied to pedicle screws to enhance osseointegration, particularly in patients with osteoporosis. As a forward-thinking exporter, we invest heavily in R&D to ensure our screw threads and surface finishes provide maximum pull-out strength and stability.
Procurement departments in modern hospitals and large-scale medical distributors have specific criteria when selecting a Pedicle Screw Systems supplier. The focus has shifted from just "price" to "total value of ownership," which includes supply chain reliability, instrument ergonomics, and post-market support.
In regions like Southeast Asia and the Middle East, there is a high demand for polyaxial screw systems that offer greater intraoperative flexibility. In contrast, European markets often emphasize minimally invasive instrumentation that integrates seamlessly with robotic navigation systems.
Our factory supports OEM and ODM services, allowing international brands to customize screw diameters, thread pitches, and rod lengths to suit local anatomical variations. This localization is crucial for ensuring that implants perform optimally across different patient demographics.
Procurement Tip: When sourcing, always verify the mechanical testing reports (ASTM F1717) for fatigue and static strength to ensure the longevity of the spinal construct.
Choosing a Chinese Pedicle Screw Systems manufacturer provides a strategic advantage in today’s competitive landscape. Our facility combines industrial-scale efficiency with artisan-level precision.
We understand that in the medical field, there is zero room for error. Our commitment to quality is backed by years of clinical feedback and international certifications, making us the preferred exporter for spinal fixation systems globally.
The company is committed to producing and selling advanced medical devices, equipment, and disposable medical supplies. Our product range primarily includes the production and sale of instruments for otolaryngology, orthopedics, urology, abdominal surgery, gynecology, and related disposable equipment, as well as sports and fitness equipment. The main focus is on orthopedic devices: implants, mechanical equipment, etc.
Our Products: Urology Instruments; Laparoscopic Instruments; Gynecology Instruments; Otolaryngology Instruments; Orthopedic Instruments; Disposable Instruments; Devices. Our team of experts is dedicated to ensuring that our products meet the highest standards of quality and safety.
Why choose Pure? Our company specializes in the production and sale of medical devices, and we pride ourselves on our commitment to excellence in every aspect of our business.
Square Meters Built
Core Licenses & Patents
High-Tech Production Lines
Exporting Countries

 Medical Equipment: Cervical Pedicle Screw System with Locking Plate
Medical Equipment: Cervical Pedicle Screw System with Locking Plate
 Disposable Titanium Spine Pedicle Screw Domino Connector Surgical Instrument Orthopaedic Implant Posterior Spinal Internal Fixation 5.5 System
Disposable Titanium Spine Pedicle Screw Domino Connector Surgical Instrument Orthopaedic Implant Posterior Spinal Internal Fixation 5.5 System
 Orthopedic Spinal Internal Fixation 6.0 System Screw Polyaxial Spine Titanium Pedicle Screws
Orthopedic Spinal Internal Fixation 6.0 System Screw Polyaxial Spine Titanium Pedicle Screws
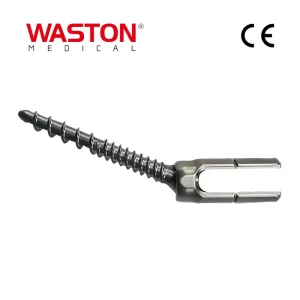 Quality Assurance Zero-X Disposable Surgical Neulen Laminoplasty Inter Pedicle Screw Orthopaedic Instrument Set Implant Spinal Fixation System
Quality Assurance Zero-X Disposable Surgical Neulen Laminoplasty Inter Pedicle Screw Orthopaedic Instrument Set Implant Spinal Fixation System
 Titanium Orthopedic Pedicle Screw System
Titanium Orthopedic Pedicle Screw System
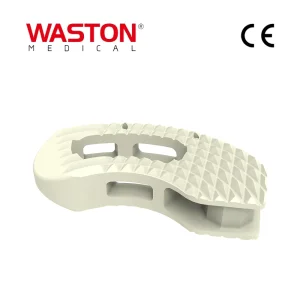 Quality Assurance Disposable Surgical Neulen Laminoplasty Inter Pedicle Screw Orthopaedic Instrument Set Implant Banana Interbody Fusion Cage System
Quality Assurance Disposable Surgical Neulen Laminoplasty Inter Pedicle Screw Orthopaedic Instrument Set Implant Banana Interbody Fusion Cage System
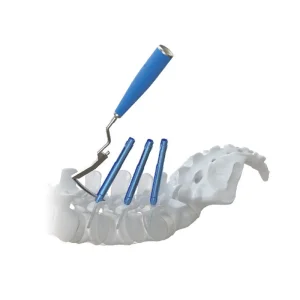 Orthopedic Pedicle Screw Spine MIS Implants System
Orthopedic Pedicle Screw Spine MIS Implants System
 OEM ODM Disposable Surgical Neulen Laminoplasty Inter Pedicle Screw Orthopaedic Instrument Implant 11127-2 Zero-X Cage System
OEM ODM Disposable Surgical Neulen Laminoplasty Inter Pedicle Screw Orthopaedic Instrument Implant 11127-2 Zero-X Cage System