




1 / 5










| Still deciding? Get samples of $ ! US$ 118/Piece Request Sample |

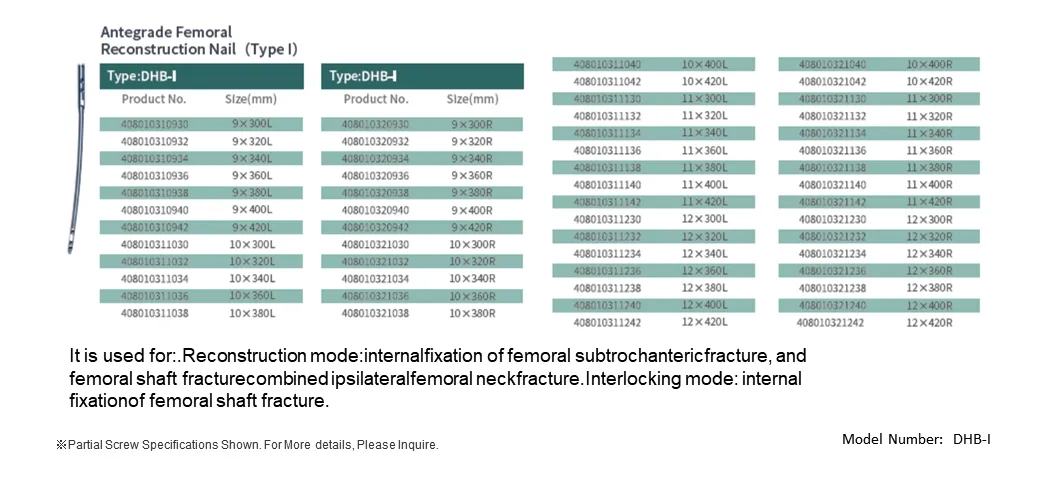
Indicated for:
As a professional orthopedic manufacturer, we provide a full product range covering trauma, spine, joints, sports medicine, and CMF — all supported by complete surgical instrument sets. Our products are designed for high-volume distribution and clinical reliability.
With in-house R&D, certified laboratories, and modern production lines, we support partners who need private label or custom solutions, focusing on reliable and scalable manufacturing for global orthopedic needs.

 Pure Orthopedic Implants
Pure Orthopedic Implants