




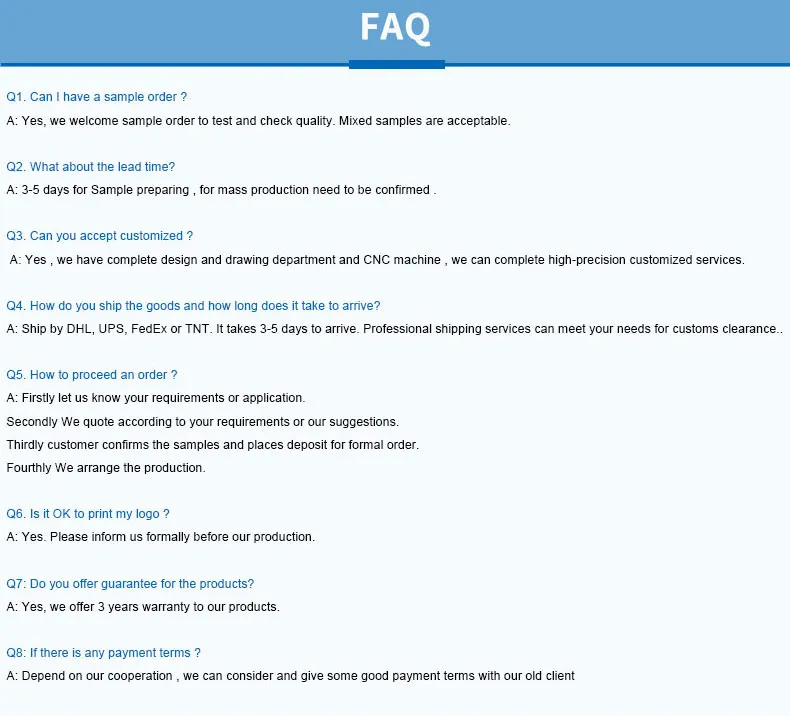
1 / 5










| Still deciding? Get samples of $ ! US$ 150/Piece Request Sample |


| Product Name | Artificial Knee Joint RSK PS Femoral Condyle Revision Knee Prosthesis Replacement |
| Material | CoCrMo |
| Color | Grey |
| Position | Femoral |
| Size | #1-8 , left/right |
| Valid Time | 5 years |
| Certificates | CE/ISO 13485 |
| Related Implant | Femoral Condyle |









 Pure Orthopedic Implants
Pure Orthopedic Implants