📋 Basic Information
Implant Type
Intramedullary Nail
Specification
Titanium Alloy TC4
🩺 Product Introduction
Medical Background: Proximal humeral fractures are among the most common fractures, accounting for 4%–5% of all cases. Usually occurs when the elbow strikes the ground or the arm is braced during a fall, or when the shoulder joint sustains direct external force.
Key Features
- Unique and innovative "nail-in-nail" technology to enhance the fixation effect of proximal locking screws.
- Proximal screws are countersunk with at least three lateral screws implanted for structural stability.
- Oblique upward supporting lock provides stable medial support for the humeral head.
- Multi-planar distal locking to avoid neurovascular structures while providing stable fixation.
- Central implantation with an ultra-low notch design to protect the supraspinatus muscle.
Screw Configuration
Ø3.5 mm interlocking screws: Can be inserted through the head of the lateral screws ("screw-in-screw") to enhance stability, particularly effective for osteoporosis patients.
Ø4.5 mm interlocking screws: Three lateral screws ensure basic structural stability. Additional anterior screws can enhance construct stability.
Ø4.0 mm ascending screw: Supports the medial calcar region, stabilizing fractures with medial comminution.
Ø4.0 mm interlocking screws: Two distal screws positioned in different planes to reduce implant toggling.
🔬 Clinical Application
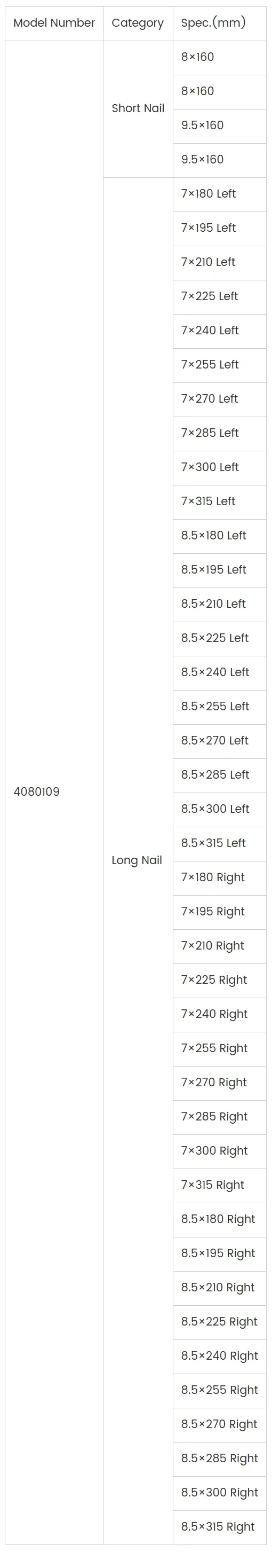
Short Nail (Length: 160 mm; Diameter: 8 mm, 9.5 mm):
- Two-part surgical neck fractures of the humerus
- Three-part surgical neck fractures of the humerus
- Four-part surgical neck fractures of the humerus
Long Nail (Length: 180–315 mm; Diameter: 7 mm, 8.5 mm):
- Proximal humeral fractures combined with humeral shaft fractures
- Humeral shaft fractures
- Impending pathological fractures
🏢 Manufacturing Capability
We are a leading manufacturer of standardized orthopedic implants and instruments, supporting global distributors and importers. Our full product range covers trauma, spine, joints, sports medicine, and CMF.
With in-house R&D, a certified lab, and modern production lines, we provide reliable and scalable manufacturing solutions, including OEM/ODM services for partners needing custom orthopedic solutions.
❓ Frequently Asked Questions
Q What shipping methods do you use?
We typically utilize courier services for shipping, but can accommodate postal services upon request. Costs are determined by destination and package weight.
Q Are your products quality certified?
Yes, our facility holds the EN ISO 13485 certification, ensuring all medical devices meet stringent safety and performance requirements.
Q What is the Minimum Order Quantity (MOQ)?
Our standard minimum order value is US$1000. However, sample orders are available at cost for evaluation before bulk commitments.
Q Can you manufacture custom or unlisted products?
Yes, we accommodate custom orders based on pictures or samples. Please note that custom production may require longer lead times.
Q Do you maintain regular stock?
We maintain stock for most catalogue items with monthly production cycles. Inventory levels vary based on current market demand.
Q Why choose your orthopedic solutions?
We are the choice of over 2,000 agents worldwide, offering standardized, high-quality implants designed for fast turnaround and emerging market needs.

















 Pure Orthopedic Implants
Pure Orthopedic Implants