








| Still deciding? Get samples of $ ! US$ 85/Piece Request Sample |
The Interface Screw is specifically designed for ACL reconstruction surgeries to secure tendon or ligament grafts in the femoral and tibial bone tunnels. It provides a robust and reliable fixation by compressing the graft directly against the bone tunnel wall. The unique mechanism ensures optimal stability and a firm attachment between the soft tissue graft and the bone.
This system includes a conical screw for easy insertion and a protective sheath that shields the soft tissue, reducing the risk of graft displacement or screw breakage. Manufactured from PEEK (Polyetheretherketone), it mimics the elasticity of natural bone, ensuring superior performance during the healing process.
The tapered shape facilitates easy insertion, with a head diameter larger than the tip to optimize placement and initial grip.
Prevents damage during insertion by enhancing holding power and ensuring the screw remains intact even under high torque.
A circular sheath shields the tendon and ligament from being cut by the screw threads, significantly enhancing surgical safety.
Features an external clamp block that keeps the graft securely in place, even under significant external pressure.
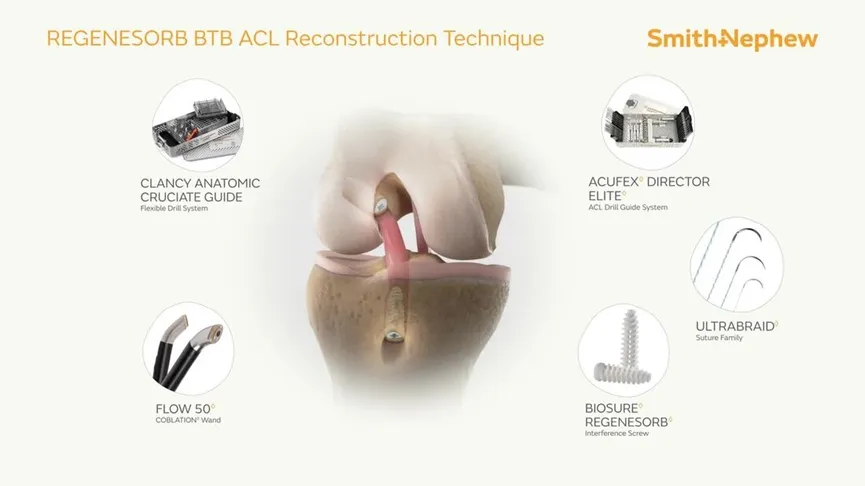

This Interface Screw System is primarily indicated for ACL reconstruction surgeries. It provides secure fixation for soft tissue grafts or bone-tendon grafts in the knee joint, promoting effective healing and functional recovery.
Professional Orthopedic Manufacturer
A leading manufacturer of standardized orthopedic implants and instruments. We offer a comprehensive product range covering trauma, spine, joints, sports medicine, and CMF. All products are supported by complete surgical instrument sets, designed for reliable manufacturing and scalability.

 Pure Orthopedic Implants
Pure Orthopedic Implants