Model NO.TY-IN
ConditionNew
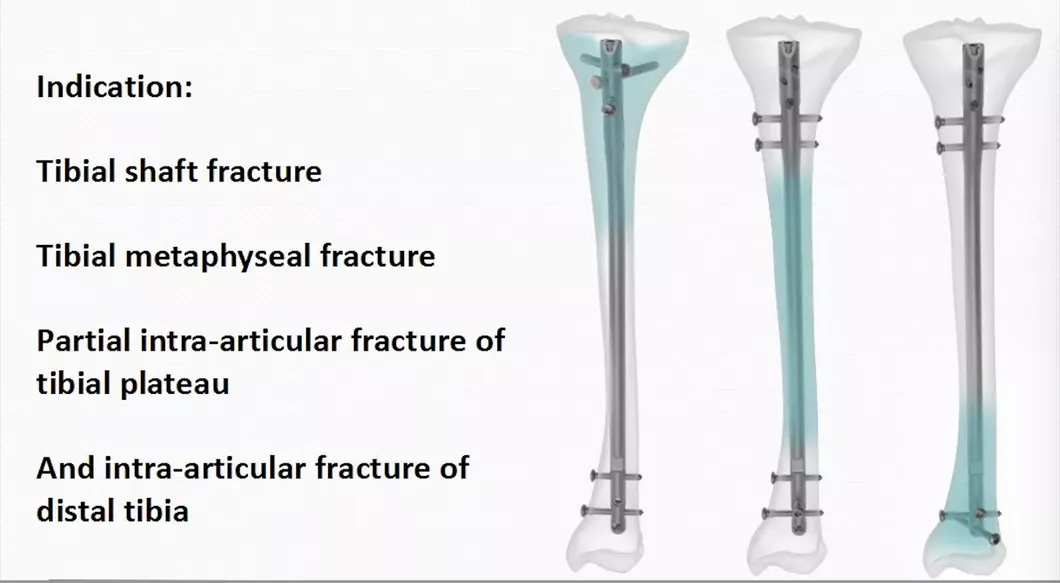
UsageCorrect Deformities, Fracture Fixation
Using SiteTrauma
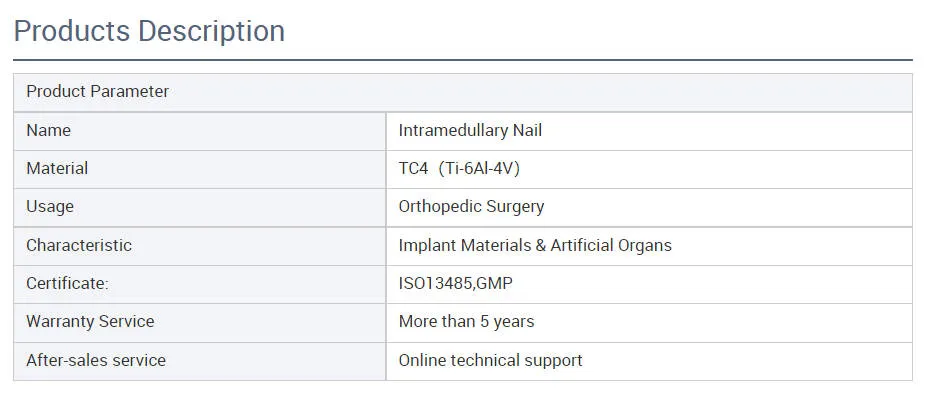
MaterialPure Titanium
ClassificationClass III
WarrantyOne Year
After-Sale ServiceOnline Technical Support
PackageSterile Package
ColorSilver
MOQ1 PCS
Licence NumberCE/ISO 13485
OEMAccept
PropertiesImplant Materials & Artificial Organs
Supply Ability2000 Piece/Pieces Per Month
HS Code9021100000
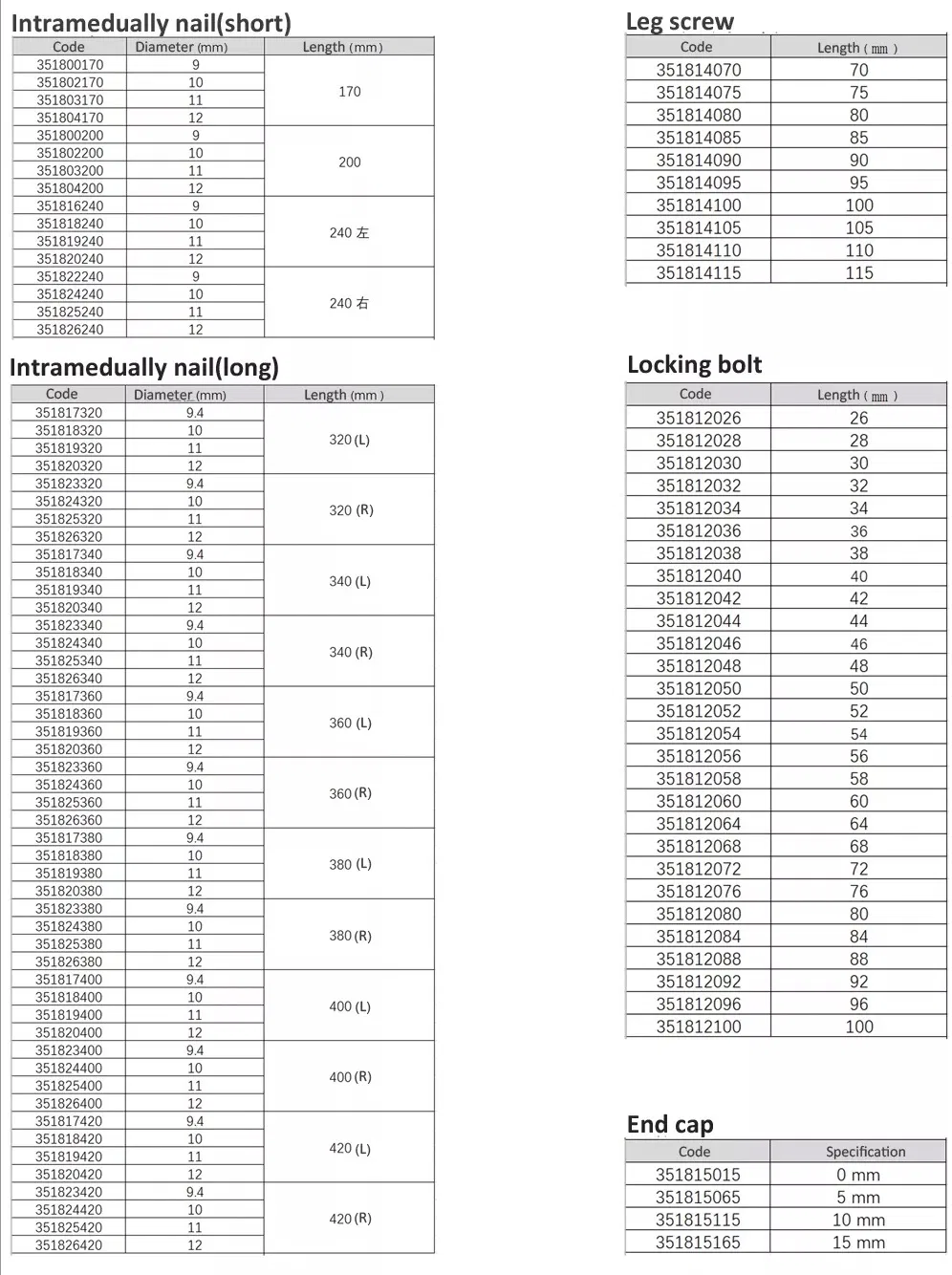
Key Features & Specifications
◈
Material: High-grade Titanium with Oxidation/Milling surface finish.
◈
Anatomical Design: Medial-lateral angle of 5° allows precise insertion at the greater trochanter tip.
◈
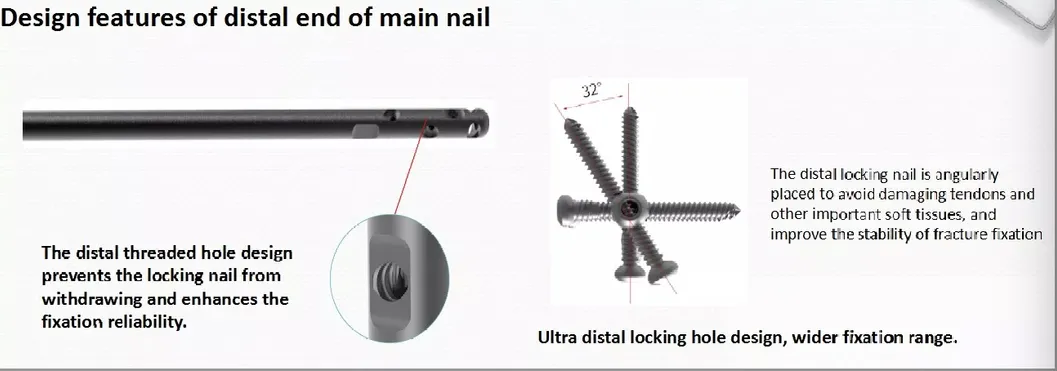
Stress Reduction: Lateral flatten design and elastic groove tip ease insertion and reduce stress to cortical bone.
◈
Curved Geometry: 240mm distal curved design prevents nail tip contact with cortical bone, reducing pain incidence.
◈
Stability: Lag screw insertion provides excellent compaction, especially beneficial for osteoporotic patients.
◈
Efficient Locking: Double thread design locking bolts to significantly reduce operation time.
What quality guarantees do you offer for the orthopedic implants?
We provide a comprehensive guarantee policy: a one-month exchange/return for quality issues (non-operational) and a one-year free repair service. After one year, maintenance is provided at the cost of parts and freight.
How do you handle technical support for these surgical systems?
We supply timeless technical maintenance service. Our engineers provide remote solutions and guidance for technical questions, allowing for efficient on-site resolutions without unnecessary delays.
Why is the price higher than some other non-branded options?
Our pricing reflects the product quality and strict medical standards. We compete on quality and reliability to ensure a win-win market outcome, rather than compromising standards for the lowest price.
Are there discounts available for bulk orders?
Yes, we consider the best possible pricing based on the specific products and quantities ordered to support long-term cooperation and profit for our partners.
What materials are used in the Intramedullary Nail systems?
The systems are primarily manufactured from Pure Titanium to ensure biocompatibility, strength, and durability required for trauma fracture fixation.
Are the products delivered in sterile condition?
Yes, the products come in professional sterile packaging, meeting the necessary medical supply standards for immediate surgical use.

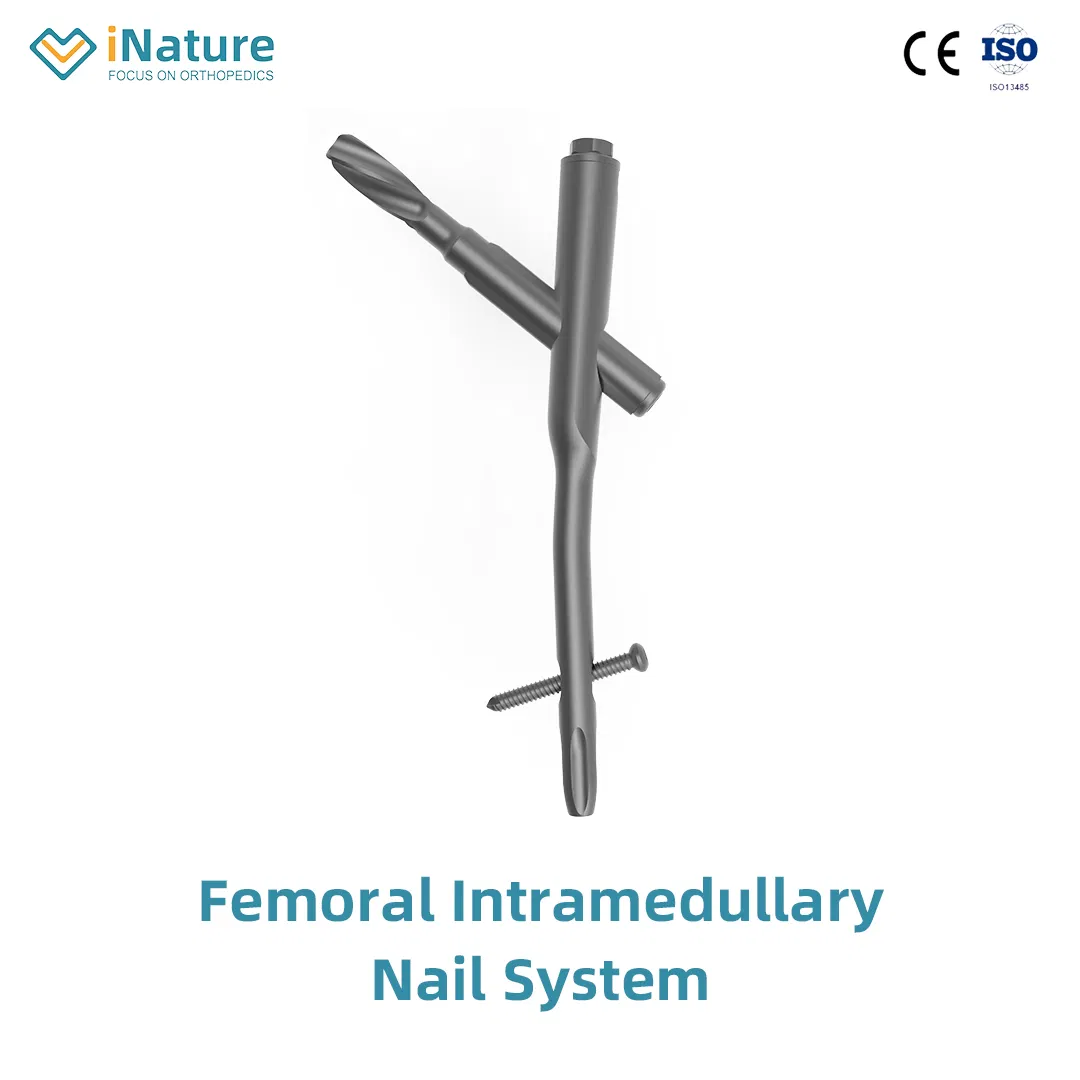



















 Pure Orthopedic Implants
Pure Orthopedic Implants