● Product Description
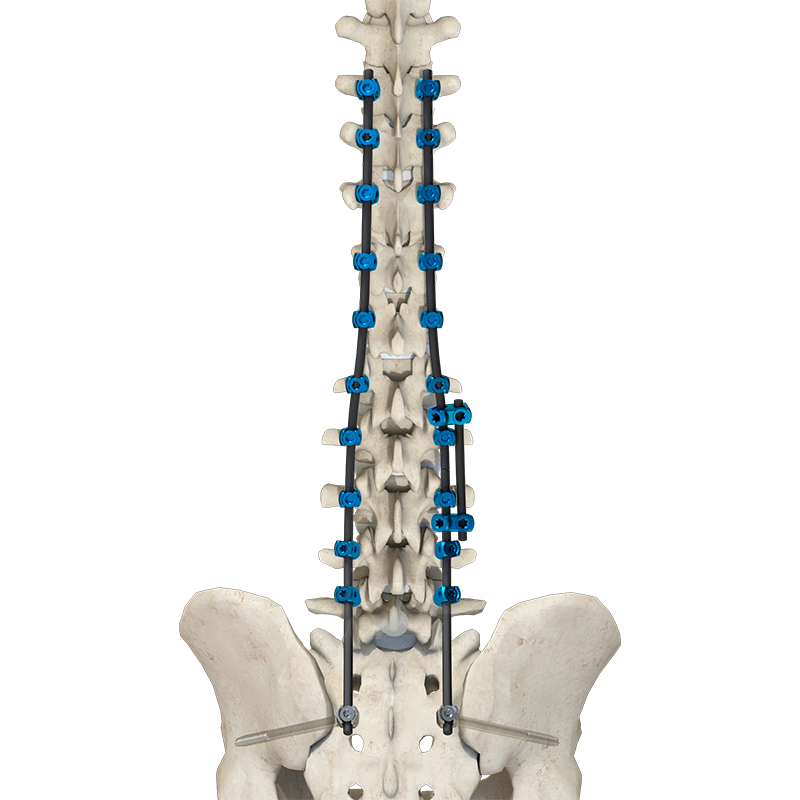
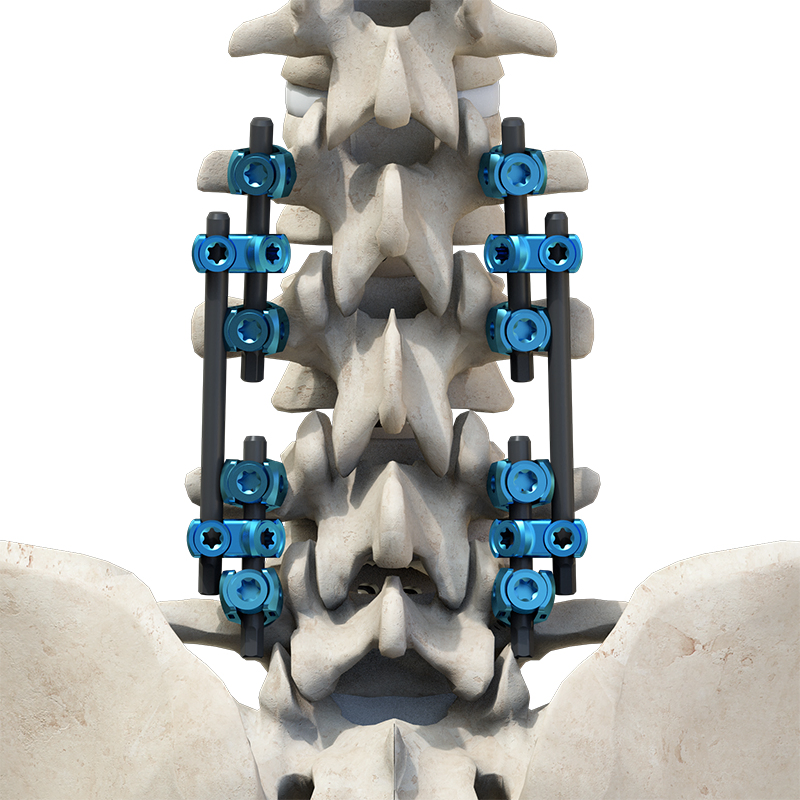
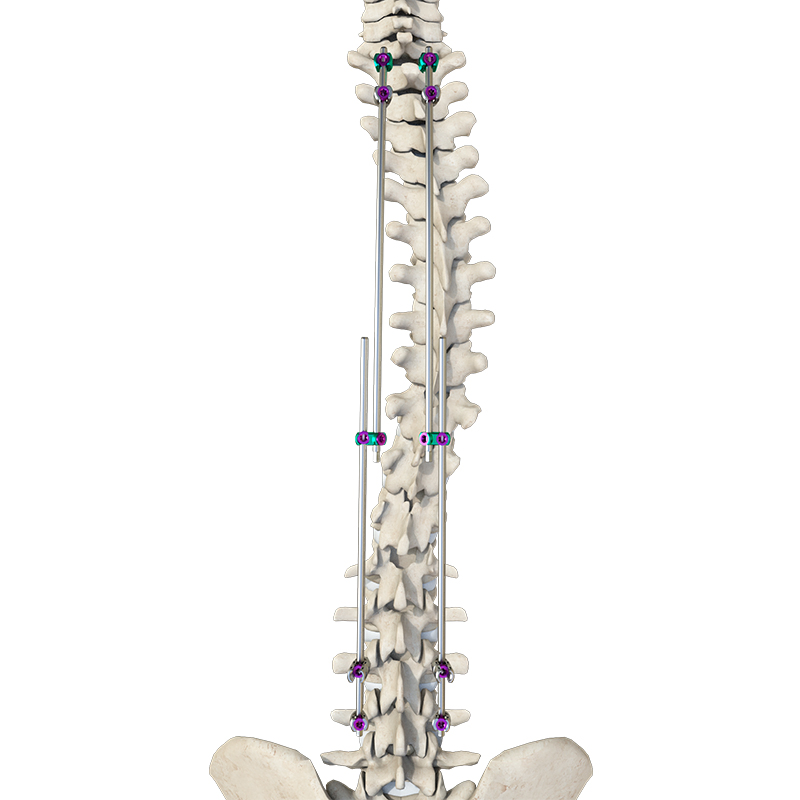
The Thoracolumbar Posterior Pedicle Screw and Rod System (CoCrMo Alloy) consists of pedicle screws, rods, set screws, crosslinks, lateral connectors, hooks, and other accessories. This system is designed to restore spinal alignment, correct deformities, and provide immediate and long-term biomechanical stability following posterior thoracolumbar procedures.
By placing pedicle screws through the vertebral pedicles and connecting them with contoured rods and various components, the system allows for distraction, compression, derotation, and realignment maneuvers. The assembly forms a stable three-dimensional construct that supports load-sharing and facilitates spinal fusion. Components are made of TC4 ELI titanium alloy, CoCrMo alloy (Co28Cr6Mo), and pure titanium (TA2G).
? Frequently Asked Questions
Are your products certified with ISO 13485?
Yes, our company holds the EN ISO 13485 certification, which is a testament to our commitment to quality and regulatory compliance in the medical device industry. This ensures our products meet stringent safety and performance requirements.
What are the shipping methods and costs?
We typically utilize courier services for shipping, but we can also accommodate postal services upon request. Shipping costs are determined by destination, package size, and weight; we provide a detailed quote once we have your specific requirements.
What is the minimum order quantity (MOQ)?
Our standard minimum order value is US$1000. However, we offer sample orders at cost to allow potential clients to evaluate our products and test the market before committing to larger orders.
Can you provide products not listed in the catalogue?
Yes, we accommodate custom orders. By providing pictures or samples of the desired product, we can work with you to create a similar item. Please note that custom orders may have a longer lead time.
Do you maintain stock for listed products?
We maintain stock for most products listed in our catalogue with regular monthly production cycles. Stock levels vary based on demand, so it is best to consult with our sales team for the latest inventory information.
Why should we choose your manufacturing services?
With over 2,000 agents choosing our solutions, we provide a stable and reliable supply chain, comprehensive R&D capabilities, and a full range of orthopedic products suitable for global high-volume distribution.
















 Pure Orthopedic Implants
Pure Orthopedic Implants